Chemistry 125
(the Registrar calls it Chem 124)
Freshman
Organic Chemistry
Fall 2010
If
you are viewing this page on Yale
Classes*V2, you should point your browser to
https://webspace.yale.edu/chem125
so
you have more space. (We use ClassesV2 mostly
for
Wiki and Schedule.)
Chem
125 lecture videos,
transcripts,
and other material from Fall 2008 are available by clicking here
(Open Yale Courses)
The videos only are mirrored here
(YouTube)
Material
for First
Quarter of the Fall Semester 2010 (lectures 1-9)
Material
for Second
Quarter of the Fall Semester 2010 (lectures 10-18)
Material
for Third
Quarter of the Fall Semester 2010 (lectures 18-28)
Material
for Fourth Quarter of the Fall Semester 2009 (lectures 29-37)
(Discussion Sections) (Exams & Grading)
Click to go to Spring Semester 2010
Do read this
More evaluations are available through http://students.yale.edu/oci/
You may want to check fall term reports for Chem 125 (2006, 2007) as well as the new name Chem 124 (2008)
Lecture 1 (9/1/2010) Viewing Instructions
Lecture for 2010 pptx 2010 Audio (begins ~4 min in) Annika movie for ppt
(2009 ppt) (2009 audio file mov) (2008 mov)
(Course Mechanics ; The BIG Question ; The Royal Society ; Pepys as a model Chem 125 student)
We'll finish up Pepys on Friday.
Note: the powerpoint file linked above is a timed file. You can run it without timing, so as to skip around by choosine "Manually" under Slide Show / Set up Show...; or you can run it in synchrony with the audio file by selecting "Browsed by an individual (window)" and "Use timings, if present". If you want to include the video clip of Annika in the powerpoint, it must be downloaded into the same folder as the pptx file.
Samuel Pepys and Isaac Newton - How might they do in Chem 125?
Note the Problems (Due Wednesday, 9/8/10)
You
may work in groups of up to 6 to prepare a group hard copy or e-mail
submission (with the names of all participants on it).
Lecture 2 (9/3/10)
2010 Timed Powerpoint pptx
2010 Audio (begins with frame 12, check 2009 audio for narration of missing slides)
(2009 ppt) (2009 audio file wma) and/or (2008 mov)
( Force Laws and Springs; Chemical Force Law? ; Lewis's Electron Octet)
You can synchronize the audio and timed powerpoint yourself, if you wish, by playing them simultaneously and using the right and left arrow keys to make the powerpoint line up with the audio.
note Problems for Friday 9/10/10 on frame 44 (Lewis structure for H/C/N in that order, H/C/H/O in any order)
Newton's Chemistry & the Business of Experimental Philosophy
Two Coulomb Problems (due Wednesday 9/8/10)
Lecture 3 (9/3/10) afternoon
2010 Timed Powerpoint pptx 2010 Audio
(2009 ppt folder zipped) (2009 Audio File WMA) and/or (2008 mov)
( Lewis Structures; equilibrium vs. resonance; acetic acid geometry & energy; Lore, O2/O3 4-D plotting; charge distribution; Earnshaw and Faraday's Lines of Force)
What's Wrong with Lewis Structures
What are Force Laws?
Earnshaw via Lines of Force
Shell Structure for Charged Balls in 2D
Dealing
with Graphs - Lesson 1
Drill
on multidimensional plotting
(you
don't need to turn the
problems in, but you should know how to do them)
answer key
(ignore that these documents are dated for previous years)
Functional
Group Table
There will be NO peer tutor session this Sunday afternoon, and no TA discussion section Monday evening.
At 10:30 Monday and Wednesday there will be small-group problem sessions with the TAs and Peer Tutors. Come to the leture room, SCL 160, at 10:30. You will be divided into small groups (according to your residential-college-based problem solving association). Two groups will stay in SCL 160, two others will meet in SCL 112, the fifth will meet downstairs in SCL 21. On Wednesday you may go directly to your discussion room.
The Monday discussion will focus on the problems to be turned in on Wednesday and multidimensional graphing;
Statistics
Coulomb's Experiments
Multidimensional Graphing
The Wednesday discussion will focus on the 8 problems to be turned in on Friday:
Lewis Dot Structures and Resonance
to which a 9th problem should be added: Compare several different Chemistry texts as to "Rules" for drawing Lewis structures and Resonance structures.
NOTE: Lecture 5 will be delivered on FRIDAY afternoon 3:30-4:30
(if you are unable to attend the live session, you may use the audio record and timed powerpoint)
Lecture 4 (9/10/2010)
2010 Timed Powerpoint (pptx)
2010 Audio
(2009 ppt) (2009 audio file wma) and/or (2008 mov)
(HCNO & Lewis structures; Plum Pudding Atoms; Was Coulomb Wrong? Clairvoyant "Science" ; Powers of 105 ; Measuring the very small - Franklin; Feeling atoms SPM)
Seeing
Atoms with Clairvoyance (relevant, but not assigned reading)
If
you're
skeptical that Crookes really believed in psychic pheomena, click here
(also not assigned, just for the curious)
SPM
web page
Also a humorous question
related to the smallness
of atoms
It will be VERY helpful to read ahead on the x-ray page
Lecture 5 (9/10/2010)
2010 Timed Powerpoint (pptx)
2010
Audio
(2009
ppt) (2009 audio file wma)
and/or (2008 mov)
(types of
SPM; seeing by wave
interference, x-ray diffraction, molecular and lattice
diffraction )
Diffraction examples from laser demonstration
Notice the "What is the line spacing?" question within ppt frame 44. You should work out an answer, so you are confident of understanding. You need not hand it in.
Lecture
6 (9/13/10)
2010 Powerpoint (ppt)
2010
Audio
(2009
ppt) (2009 audio file wma)
and/or (2008 mov)
(understanding
molecular and lattice
diffraction, Franklin's DNA photo; e-density maps, difference
maps and bonding)
Leslie
Leiserowitz on his Triene (Transcript 16K)
Leiserowitz Quicktime Movie
(160 x 120, 5.6 Meg) or (240
x 180, 10.3 Meg)
Dunitz Quicktime Movie (160 x 120, 6.2 MB) or (240 x 180, 11.2 MB)
(You are not responsible for the page on anomalous dispersion.)
The Second Key Question
It will be VERY helpful to read ahead on the Quantum Mechanics page
Lecture 7 (9/15/10)
2010 timed Powerpoint zipped with Schrödinger sound (ppt) or (ppt only)
2010 Audio
(2009 ppt downloads zipped folder containing ppt and Schroedinger sound files) (2009 audio files wma 1 frames 1-18, wma 2 frames 19-30 ) and/or (2008 mov)
(Pathological Bonding; Schroedinger's Invention; Weird Kinetic Energy; The Jeopardy Method, Goldilocks)
Quantum Mechanics (Sections I-V on 1st Exam)
Erwin Meets Goldilocks (problem set due Monday, Sept. 20 - PDF document)
|
Jon will demonstrate the use of Erwin Meets Goldilocks at the Thursday discussion section. This program will help you develop a quantum mechanical intuition. You will be doing the relevant problem set in self-selected groups. Be sure that at least one representative from your group (or someone else who is willing to help you) attends the demonstration session. Bring your laptop to the session. It would help if you download the Applet ahead of time. |

thanks to Yoonjoo Lee
|
Lecture 8 (9/17/10)
2010 Powerpoint (ppt)
(I apologize that, by mistake, the link was not updated until long after the ppt itself was online. I try to update the ppt and make the audio available soon after the lecture. Please do let me know in such a situation, and try changing the address manually in your browser to the current year.)
2010 Audio
(2009 ppt) and/or (2008 mov, see also a transcript) (no 2009 audio file because of operator malfunction - sorry)
(1D Quantum - Erwin meets Goldilocks; Nodes & Quantization; Ψ2 & Structure; Vibration, Double Minima)
Lecture 9 (9/20/10)
2010 Powerpoint (pptx)
2010 Audio
(2009 ppt) ( 2009 audio WMA) or (2008 mov, see also a transcript)
(Double Minima - bonding and tunneling; Chladni - nodes in 2D; H-like atom wave functions; ρ for r, orbital shapes)
Chladni Figure Short Movie (Quicktime - 3MB)
4 Chladni Figures (Quicktime - 9.5 MB)
Quantum Mechanics V: H-like Atoms & problem set
Problems
for Wednesday, September 24 :
2)Why are the first two cells [(0,0) (1,0)] in Chladni's tables vacant?
3) Compare 1s of H with 2s of C+5 in Energy
4) Do the six Atomic Orbital problems HERE (also for Atom-in-a-Box Download)
Clicking here links to a Java program that provides some of AiB functionality for a PC.
Note: AiB will not be covered on Friday's exam
The exam at class time on Friday, Sept. 22, will cover through Lecture 9 (Lecture 10 will be covered on the next exam)
60 minutes will be alloted for the exam, either 10:30-11:30 in SCL 160 (the normal lecture room) or 10:15-11:15 in SCL 111 down the hall. That is, you can either stay 10 minutes late or come 10 minutes early.
Prof. McBride will lead a review session on Wednesday, Sept. 20, in room 119 WLH from 8 to 10 pm. Come with questions.
Note incidentally that the Monday and Thursday discussion sections are held in WLH 204 (contrary to what the syllabus used to say - sorry for the mistake)
You may be wondering what kind of questions might appear on exams in this course. (Our first exam will occur on Friday, Sept. 24 The following PDF file contains a number of questions based on topics we have been discussing. Most of these would be fair game, but a few from earlier years refer to material we won't cover until shortly after the first test. It might be useful to look them over as we go along.
Sample questions for first hour exam
Sample Quantum Mechanics Questions (PDF)
Sample questions on Early Topics (PDF)
First Hour Exam 2010 (pdf) First Hour Exam 2010 Answer Key(pdf)| Exam
scores are
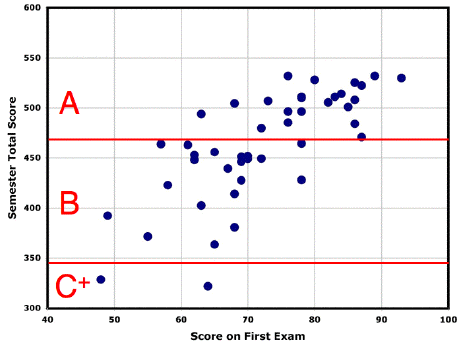
available via Post'em. The class average was very good (75.6) with 1/3 of the scores greater than 77 and 2/3 of the scores greater than 71. Congratulations, you seem to be learning a lot! (although some show need for improvement in drawing resonance structures) You can check your own points, both by question and total, using Post'em on ClassesV2. If you have questions about the grading, consult the answer key (above) first, and then speak to the grader of the relevant question (1-6, Miller; 7-10, McBride). These scores are of course almost insignificant, because they represent such a small, preliminary portion of the grading for the semester. But the course content is cumulative, so it can be helpful to see where you stand thus far, and whether it is important to be sure you're not falling behind. There is plenty of help available from the teaching staff. 5/6 of the semester scoring is still to come, so it's way too early either to despair or to become self-satisfied. The plot to the right from several years ago shows that the first exam score was not a great predictor of semester total. There is some correlation, but it is easy by working (or not) to move up (or down) by a letter grade. |
 |
Material for the second quarter of the Fall Semester 2010
Lecture 10 (9/22/10)
2010 zipped timed Powerpoint (pptx) (folder includes juggling video) (or pptx only)
2010 Audio
(2009 ppt - downloads zipped folder with ppt file) ( 2009 audio WMA) and/or (2008 mov, also here is a transcript)
(Parts of 2p; Dauger's Atom-in-Box; r2 weighting; scaling size and energy; hybridization for orientation and shape; The Problem With Orbitals)
If you
are have difficulty with why r2 is
sometimes used to multiply Ψ2
in
discussing electron distribution,
you might try these optional
problems from a previous year and discuss them with friends
or TAs.
Lecture 11 (9/27/10)
2010 Powerpoint (ppt)
2010 Audio
(Lecture 11 ppt) (2009 Audio wma) or (2008 mov)
( 2-electrons, Zeff, SCF, "Correlation Energy", Energy Magnitudes; Orbital Shapes; "Plum-Pudding" molecular orbitals)
Quantum Mechanics VI-VII
(orbitals for several electrons, hybridization)
Energy Scales and Correlation Energy
Lecture 12 (9/29/10)
2010 Powerpoint (ppt)
2010 Audio
(Lecture 12 ppt) (2009 Audio wma) or (2008 mov)
Quantum
Mechanics VIII-IX
(molecular orbitals; HOMOs and LUMOs for functional groups)
H-H Bonding
C-C
Overlap and Hybridization
Lecture 13
(10/1/10)
2010 Powerpoint (ppt)
2010
Audio
(Lecture
13 ppt)
(2009 Audio wma
or
(2008 mov)
(Newton's Grail;
Energy-Match, H2 vs. HF, hybridization &
geometry; Reality; Predictions for Experimental Test with XH3)
XH3 (contact with reality)
Lecture 14
(10/4/10)
2010 Powerpoint (ppt)
2010
Audio
(Lecture
14 ppt)
(2009 Audio wma
1st 25 min only, sorry)
or
(2008 mov)
(XH3:
IR, ESR, CF3; Goals;
Pathological
Bonds; BH3
and
local bonds)
BH3,
an Example of an MO Computation & an Apology for Local Bonds
Note:
the pictures on this page are fun to look
at, but
don't
worry too much about the verbal content,
unless you can't sleep because we use local bond analysis instead of
the MOs that computers find.
Lecture 15
(10/6/10)
2010 Powerpoint (ppt)
2010
Audio
(Lecture
15 ppt)
(2009 Audio wma)
or
(2008 mov)
(BH3
and
local bonds; Which
mixings are important?; Acid-Base Theory; What makes HOMO high, LUMO
low?)
MO
Interactions, which ones count? (HOMO/LUMO)
Lecture 16
(10/7/10)
2010 Powerpoint (ppt)
2010
Audio
(Lecture
16 ppt)
(2009 Audio wma)
or
(2008 mov
& mov)
(What
makes HOMO high, LUMO low? B2H6;
HF
LUMO; Acid-Base
- SN2 - E2 LUMO Analogies;
Make &
Break)
|
|
|
|
|
|
|
|
LUMO Analogies (elimination from C2H5X)
Last year pairs of class members adopted a functional group to discuss in terms of pair-wise and atomic orbitals (and their interactions). You should study their Wiki pages, decide whether you agree with their analyses, and be prepared to produce such a report on any of these functional groups, or on some unfamiliar functional group.
If you would like optional problems to help firm up your understanding of quantum mechanics before the exam a week from Monday (Oct. 18), you could look at the exams from previous years linked below.
Lecture 17
(10/8/10)
2010 Powerpoint (ppt)
2010
Audio
(Lecture 17 ppt)
(2009 Audio wma)
or
(2008 mov
& mov)
(N2H4
Synthesis;
Four
Functional Groups: Carbonyl-Buergi-Dunitz
Angle, Amide)
Note: the last half-hour of the 2009 wma recording (beginning at 57:30) contains the optional after-class demonstration of Spartan in which MOs of an enol were calculated and compared with class-member predictions. After the END frame of the Lecture 17 ppt, there is a supplementary timed ppt showing some screen shots from the demonstration. This is optional, not required reading/listening, but it may help to make the computer's contribution less vague.
Where should R2C=O be attacked? (Buergi-Dunitz Angle)
Intramolecular HOMO/LUMO mixing and
"Resonance"
]
(not to turn in, but you may want to discuss it)
Lecture 18 (10/11/10)
2010 Powerpoint (ppt) link corrected to 2010
2010 Audio
(Lecture 18 ppt) (2009 Audio wma) or (2008 mov)
(Functional Group Analysis: Amide cont., Carboxylic Acid, C-Metal bond and aggregation. How Did They Know? Precursors of Modern Chemistry- Alchemy)
3-Center "Electron-Deficient" "Y" Bonds
[ B2H6 & (CH3Li)4 •4 OMe2
History (how humans actually learned about organic chemistry)
Why we study chemical history (DO READ THIS & the others)Your Genealogy in Organic Structure Theory
Review Sessions for the Exam on Monday, Oct. 18
Audio (.mov Quicktime file) of McBride review session Tuesday, Oct 12
(of limited utility without gestures and blackboard, but here it is)
The Peer Tutors and Graduate TAs will hold their regularly scheduled help sessions on Monday and Thursday evenings and Sunday afternoons. In addition Jon will hold a help session on Friday morning, Oct 15, 10:30-11:20 in 160 SCL (the normal lecture time and place). These are the opportunity for you to practice functional group analysis.
Note: the exam will focus on Lectures 10-18 but will not include the history material from the last 21 minutes of Lecture 18
Sample Problems for Oct. 18 Exam
The exam will take place at 10:30 AM (normal lecture time) in SCL 160 (normal lecture room). It will be budgeted for 50 minutes, but 60 minutes will be allowed for writing exam papers. To accommodate students who must leave for the following class (or who just wish to get it over with), the exam may be taken early from 10:15-11:15 in SCL 111 (next to southeast corner of SCL - at other end of the hall from 160).
2nd Hour Exam 2010 (pdf) 2nd Hour Exam 2010 Answer Key (pdf)
Material for 3rd Quarter of First Semester 2010
(Alternate source for 3rd Quarter Links)
Lecture 18 timed (ppt) 2010 Audio
The material after slide 14 (Audio 30 min) is for this 3rd quarter of the course
(Yale chemistry and equipment 100 years ago and its alchemical roots; the Doctrine of Sympathies; Scheele's Laboratory)
Lecture 19
(10/13/10)
2010 Powerpoint (ppt) 2010
Audio (first
2.5 min. exclamations about Hg)
(Lecture 19 ppt)
(2009
Audio wma)
or
(2008 mov)
(Scheele's
Acids; Oxygen; Lavoisier's
Revolution:
Nomenclature, Traité, Analysis, Elements,
Measuring Gases, Radicals & Language, Measuring
Heat)
NOTE: This lecture will be covered on subsequent exams, not this exam. Concentrate on quantum mechanics (and Lect 18).
Prerevolutionary
contributions - Scheele
(14 Questions for Friday, Oct 22, indicated by * below (you may work in groups)
Portrait (we discuss the problem re. Fig. 10 in class, you need not turn it in)for no reason that I can understand the first link below works only when you click the first C
Combustion of Phosphorus and Carbon*4
Quantitative Chemical "Formulae" *4
1789 Preface
Elements & Oxidation *2
How to determine Gas Density
Calorimeter *4
Lecture 20 (10/20/10)
2010 timed Powerpoint (ppt)
2010 Audio (wma)
(Lecture
20 ppt)
(2009 Audio wmv)
or (2008 mov)
(Lavoisier
Analysis
& Bookkeeping; Dalton: Atoms &
Proportions;
Berzelius: Symbols & Analysis; Gay-Lussac
Volumes; Mitscherlich
As/P ratio; )
Combustion Analysis Lavoisier/Prout/Liebig/Dumas (1788-1841)
Note:
problems for Wednesday,
October 27
(on web pages with red
years 1828 and 1832 below) Liebig's Kaliapparat and
the etymology of
K and Na (1831) 1828-1851
Isomerism
Distillation and the
"Liebig"
Condenser (1771)
Radical (Dualistic) vs.
Type (Unitary, Substitution) Theory
Woehler's
Urea Paper (1828) (analysis
and isomerism preview)
Berzelius Coins the Term Isomeric
(1830)
Woehler/Berzelius
on
Liebig, Isomerism, & Organic Chemistry (1830-35)
Woehler/Liebig
Discovery of
Benzoyl Radical (1832) (by
analysis)
Lecture 21 (10/22/10)
2010 timed Powerpoint (ppt) 2010 Audio (wma)
( Berzelius: Analysis, Electrolysis & Dualism; Woehler & Urea; Isomerism; Liebig Analysis)
Lecture 22 (10/25/09)
2010 Powerpoint (ppt) 2010 Audio (wma)
(Lecture 22
ppt)
(2009 Audio wmv)
or (2008 mov)
(Radical
Theory; Addition and Substitution Reaction Mechanisms; Type Theory; Valence: Couper)
Dumas's Panegyric on the
Radical
Theory (1837)
Berzelius on Dumas
Substitution
Theory (1838)
Simple Alkane Etymology
Woehler's
Spoof of the Type
Theory (1840)
Competing
Views of Organic Chemistry (~1851)
1858-1860s
Constitution
"Nature and Sequence of Bonds"
Couper
On a New Chemical Theory
(1858)
Kirkintilloch Movie (11Mb)
Kekulé
on the Superiority of his Sausage Formulae
(1865)
Other Molecular Diagrams
and Models
(mid 1860s)
Cannizzaro, the
Cautious
Revolutionary,
on Models (1872)
Lecture 23 (10/27/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(2009 Lecture
23 ppt
) (2009 Audio wmv)
or (mov)
(Valence: Couper, Kekulé; Notation, Models,
Counting Isomers)
Note isomer counting Problem
for Friday on Frame 48 (if we get that far, otherwise for Monday)
"Arrangement of Atoms in Space"
Koerner Proves 6-fold
Symmetry of
Benzene
(1869)
Paternó's
Tetrahedral
Carbons
(1869)
Lecture 24 (10/29/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(Lecture 24 ppt
) (2009
Audio wmv) or
(2008 mov)
( Koerner's
Benzene Proof, Paterno's tetrahedral carbon)
Lieben to
Paternó on Atoms
in Space
(1869)
Lecture 25 (11/1/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(Lecture 25 ppt ) (2009 Audio wmv
- sorry, only first 20 min) or
(2008 mov)
(Benzene
Structure - van't
Hoff vs.
Ladenburg; Carvone/Tartaric Acid Isomerism; Pasteur, van't Hoff)
van't Hoff's Tetrahedral
Carbon
(configuration) (1875)
Kolbe's
Criticism of van't Hoff
(1877)
Your
Teacher, a Later-Day Kolbe?
Lecture 26 (11/3/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(Lecture 26 ppt
) (2009
Audio wmv) (2008 mov)
(van't
Hoff,
Tetrahedral C; Coordinate Transformation; Alkenes & Allenes;
Mirrors
& Alice; Chirality;
Stereochemistry; Notation; Fischer Counts Configurational Isomers)
Lecture 27 (11/5/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(Lecture 27
ppt) (2009 Audio wmv) (2008 mov)
(Fischer's Ambiguous DL Nomenclature for
Configuration. Leiserowitz on Absolute Configuration and the Stereochemistry of Malaria)
Using Asymmetric Crystal Growth to Identify Molecular Configuration
Higher on this same webpage is an explaination of Bijvoet's anomalous dispersion technique for which you are NOT responsible.
Lecture 28 (11/8/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(Nomenclature for constitution and configuration)
AUDIO
OF THE LAST 2/3 OF THE MONDAY REVIEW SESSION (OF LIMITED INTEREST,
CERTAINLY BETTER TO SPEND YOUR TIME ON THE LECTURES AND OLD EXAMS)
Sample
Nomenclature Drill (Yale only)
3rd
Hour Exam 2004 (pdf) 3rd
Hour Exam 2004 Answer Key (pdf)
Old Third
Exams for Practice :
Note - we
are
ahead of the pace for several previous years and very slightly ahead of
last year,
so different
material covered in some of the earlier exams will be relevant this
year.
3rd Hour Exam
2003 (pdf) ...3rd
Hour Exam 2003 Answer Key (pdf)
3rd
Hour Exam 2006 (pdf)
3rd
Hour Exam 2006 Answer Key (pdf)
3rd
Hour Exam 2007 (pdf)
Sorry, no answer key
(even
earlier exams available in the table below)
The
exam will be offered 10:15-11:15
in 111 SCL and 10:30-11:30
in 160
SCL
3rd
Hour Exam 2008 (pdf)
3rd
Hour Exam 2008 Answer Key (pdf)
3rd Hour Exam 2009 (pdf) .3rd Hour Exam 2009 answer key (pdf)
Exam Score
Statistics 2009
Mean
1/3 of scores
>
2/3 of scores
>
3rd Exam
84.2
88
82
Sum of 3 Exams
234.7
247
222
3rd Hour Exam 2010 (pdf) Answer key not ready yet
Material from 4th Quarter of Fall Semester 2010
Lecture 29 (11/12/10) Prof. K. B.
Wiberg guest lecture
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
(2009 mov
or mp4)
(2009 audio wma)
(2008 Laurence D. Barron on Optical Rotation mov)
(Wiberg describes some of his contributions to classical configuration correlation and the calculation of optical rotatory strength)
Stereochemical
Nomenclature:
Cahn-Ingold-Prelog
Chirality in the 8th
Century
(note Problem for Monday)
Problem
for Monday Nov 15 (based on frames 17-18 of Lecture 27)
In
1927 P. A. Levene et al. used reaction with HI to convert
butane-1,3-diol into 4-iodobutane-2-ol and thus related their
stereochemical configurations. In 1952 young K. B. Wiberg
worried
that an alternative mechanism for this conversion might invalidate
Levene's conclusion. Use HOMO-LUMO analysis for possible
reactions to see what worried Wiberg. At each reaction step
consider alternative possibilities for reaction. (You may
work in
groups on this problem.)
Lecture 30 (11/15/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wma)
( Optical Rotation, Resolution;
Aldol Reaction; Enantiospecificity)
Lecture 31 (11/17/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2008 Dr. Dianne Duffey presentation on clinician perspective)
(Stereo
Viewing; Who Cares Drugs; Esomeprazole:
name, chirality, mode of action, Chiral Switch, Clinician Perspective,)
(Stereo Viewing this is optional, ask if you're interested)
Thalidomide/Ritalin Chiral Switch
Lecture 32 (11/19/09)
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio wma) (2008 mov
and
mov)
(Litigation; Resolution
& Chiral Synthesis,
Sharpless Allylic Oxidation; Omeprazole Mechanism; Conformation
&
Energy; Newman Projection)
Lecture 33 (11/29/10) 1885-
Conformation & "Strain"
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio wma) (2008 mov
)
(Rotational Barrier from Heat Capacity; Source
of Barrier; Stereotopicity and Toponomy; Baeyer Strain Theory)
Topicity: Prochirality and
Dehydrogenase
Sachse Identifies Conformational Isomers; Baeyer objects (1890-93)
Lecture 34 (12/1/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio wma) (2008 mov
and end of mov)
(Sachse's
Rhetoric; Mohr's Drawings; Drawing
Cyclohexane; Barton's Conformational Analysis; Ring Flips; Rotational
Barriers; Strain Energy and Molecular Mechanics)
Mohr Vindicates Sachse (1918)
Drawing Perspective Views of Cyclohexane
Lecture 35 (12/3/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio MP3 N.B.
this is an MP3 file because I had difficulty editing the wma. Tell me
if you have trouble with it.) (2008 mov )
(Axial
vs. Equatorial
Energy; Ring Relaxation; MM vs. Quantum; )
Lecture 36 (12/6/10)
2010 Timed Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio wma) (2008 mov )
(Generic
Geometries and CSD; Energy
Models; Heat of Formation;
Average Bond Energies; deltaHatomCarbon,
"effects" in ketone-enol equilibrium; Statistics and Equilibrium)
Energy
Energies:
Choice of Zero and Chupka's Heat of Atomization of Carbon
Lecture 37 (12/8/10) Relevant websites:
2010 Powerpoint (ppt) 2010 Audio (wmv)
(2009 ppt) (2009 audio wma) (2008 mov )
(Statistics
and Equilibrium; Reaction on the Potential Energy Surface; why we studied The
Chemical Bond)
Boltzmann
Distribution
Distributions
- Maxwell to Bush (powerpoint) (marginal
relevance)
Entropy
& Disorder Riddle
REVIEW SESSIONS: Thursday, Dec 9, 7-9 pm, WLH 204, Miller Friday, Dec10 10:30-11:30 am, 160 SCL, McBride Sunday, Dec 12, 3-5 pm, WLH 117, Peer Tutors Monday, Dec 13, 7-10 pm, WLH 204, Miller Wednesday, Dec. 15,
8-10 pm, WLH 208, McBride Thursday, Dec. 16, 7-9 pm, WLH 204, Lichtor & Miller
Audio Recording (wmv)
The beginning of this session concerned exam struture (0:00-1:30), then an excursion through recent topics (5:00-14:00)
and some thermodynamics/statistics (H 14:00-22:30; S 22:30-32:00)
Much
of the conversation turned on understanding how heat capacity and
entropy could relate to the rotational barrier in ethane (Lecture 33
slides 4-7)
(33:30-55:30 1:05:00-1:16:30, 1:17:50-1:29:00) We have now spent more than enough time on this topic.
Other
topics: numbers worth memorizing (59:30-1:00:10); configurational
nomenclature (1:00:10-1:04-50); utility of models (1:16-30-1:17:14)
Audio Recording (wmv)
(Note that it may be more useful to spend time reviewing the
organized lectures than this stream-of-consciousness session)
Topics
covered included: Stereochemistry-Resolution (00:28-8:00), tartaric
acid (8:00-16:30), nomenclature, history of the 4Cs (16:30-29:00);
Thermodynamics,
Statistics, Transition State (29:00-44:30); Chiral Catalysis
(44:30-52:00); Reaction Mechanisms (52:00-1:06:00);
Fischer Projection (1:06:00-1:10:00); Seeing Molecules, SPM, X-ray (1:10:00-1:18:00);
Quantum
Mechanics (1:18:00-1:23:00), Psi(x) to V(x) (1:23:00-1:30:30),
Floppiness vs. number of quantum states (1:30:30-1:34:15);
Reaction
Coordinate, Potential Energy Surface, Transition State Theory
(1:34:15-1:39:20); Topicity, Conformation, and Configuration
(1:39:20-1:52:00)
9
AM - 12 Noon Friday, December 17 SPL 59 Previous
Fall Final Examinations: Note:
Some
previous
exams covered a
few more topics that we did not cover this year. You are
responsible
only for what was presented in lecture this year and for assigned
reading and
problems. 1999 First Semester Final
Exam
(pdf 128kB) and Answer
Key
(pdf 68kB) 2000
First Semester Final Exam
(pdf 180kB) and Answer Key (pdf 48kB) 2001 First Semester Final
Exam
(pdf 180kB) and Answer
Key
(pdf 220kB) 2002 First Semester Final
Exam
(pdf
164kB) Answer
Key
(pdf 192kB) 2003
First Semester Final Exam (pdf 380kB)
(sorry no answer key) 2004
First Semester Final Exam 2005
First Semester Final Exam (sorry no answer key) 2006
First Semester Final Exam
Answer Key
Supplemental information for above:
Resonance Structures
ProtonatedMeOHLUMO
2008
First Semester Final Exam Answer Key
2009 First
Semester Final Exam Answer Key
Question
12 ribosome movie
Material for First Quarter of the Fall Semester 2009
Material for Second Quarter of the Fall Semester 2009
Material for Third Quarter of the Fall Semester 2009
Material for Fourth Quarter of the Fall Semester 2009
|
|